It happened again that morning. During their rounds, zookeepers found another tank of dead blue poison dart frogs. The tiny azure amphibians, native to South American rainforests, had been enjoying a successful breeding program at the Smithsonian National Zoo. Now, inexplicably, they were dying from a mysterious skin disease and the cause remained elusive.
The year was 1996 and Allan Pessier (’96 DVM) had just begun a pathology residency at the National Zoo. As a lifelong amphibiophile, he was more than a little intrigued when the deceased dart frogs began arriving in his laboratory.
Together with senior pathologist Don Nichols, Pessier used an electron microscope to search for the likely culprit. It wasn’t long before they zeroed in on what appeared to be an unusual fungus called Chytridiomycetes or “chytrid” that typically grows on decaying vegetation.
Seeking verification, Pessier used their pre-Google web browser to locate one of the world’s few experts on chytrids, a mycologist named Joyce Longcore. Longcore agreed their specimen seemed to be a chytrid but was unlike anything she’d ever seen before. Eventually, she identified it as an entirely new species and the first chytrid fungus known to infect vertebrates like frogs. They named it Batrachochytrium dendrobatidis or Bd.
Around the same time, researchers in Australia and Central America announced the discovery of a protozoan they believed was causing the ominous global decline in wild frogs that had been occurring since the 1970s. Incidents of these massive die-offs had risen sharply in the 1980s but no one could pinpoint exactly why.
When Pessier’s team saw photos of the suspect microbe in the New York Times, they immediately knew the organism was not a protozoan, but was instead Bd, the chytrid fungus they had just identified. With a dawning awareness, they recognized their discovery could have enormous implications.
“We thought we’d discovered a cool thing in zoo frogs,” says Pessier. “But, there was this window of time when we realized we may be the ones who knew exactly what was causing these enigmatic global die-offs. That’s pretty exciting, especially when you’re just a year out of veterinary school.”
In time, their theory was validated and Pessier began providing diagnostic help to investigators in Central America. Since Bd thrives in a cool moist climate, frogs living in mountainous cloud forests suffered the most, particularly those in Panama like the iconic golden frog now thought to be extinct in the wild.
“It didn’t hit me how devastating it really was until I went out into the Panama rainforest in 2006,” Pessier says. Before the chytrid fungus went through, the forest was deafening with frog calls and they covered the ground everywhere you stepped. But after the chytrid fungus, it was completely silent and you had to search for 45 minutes to find a single frog.
“Bd has truly earned its name as the most deadly pathogen ever recorded,” he says, speaking of the worldwide analysis published last March in the journal Science. The report concluded that Bd, unrivaled in its ability to kill untold hundreds of millions of frogs, was responsible for the decline of more than 500 species with at least 90 forced into extinction. Deadlier than the 1918 influenza pandemic or medieval bubonic plague, Bd is the worst infectious disease known to science.
In 2017, Pessier, Nichols, Longcore, and colleague Elaine Lamirande were honored with the Golden Goose Award from the American Association for the Advancement of Science. The award is given to groups of researchers whose seemingly obscure, federally-funded research has led to major breakthroughs in medicine, science, technology, the environment, and more.
Today, in an office adorned with frog paintings and posters, Pessier has returned to Washington State University as a pathologist in the Washington Animal Disease Diagnostic Laboratory (WADDL) and clinical associate professor in the College of Veterinary Medicine. With 20 years’ experience in aquatic pathology, Pessier is the “go to guy” when zoos and other organizations have tough questions about amphibian disease. Each year, WADDL receives hundreds of samples from people across the world seeking Pessier’s expert knowledge and diagnostic skills.
But Pessier is just one of several WSU scientists taking amphibian research to the global level. Caren Goldberg in the School of the Environment is a pioneer in the development of environmental DNA (eDNA) techniques that simplify the ability to screen for pathogens like Bd that can be spread through the international pet trade.
Other researchers in the School of Biological Sciences are investigating physiological and environmental stress factors that could help trigger mass amphibian die-offs. Their findings have applications for many other species as well.
Together, this diverse group of scientists has created a synergy that puts WSU in the national spotlight as an emerging center for amphibian research. They share a critical goal: To prevent the occurrence of a second fungal pandemic—an explosive threat looming just over the horizon.
Rain clouds are gathering but, for the moment, it’s a sunny April afternoon as I follow a muddy path around the little pond at Virgil Phillips Farm Park just outside Moscow, Idaho.
Making my way through trees and cattails, I join assistant professor Goldberg, who is busy assembling her eDNA collection system. Dressed in jeans and tall rubber boots, she kneels in faded grass near the edge of the water where two male Columbia spotted frogs have staked out territories.
Goldberg quietly lowers a plastic tube into the pond and uses a hand pump to draw water up through a filter and into a flask. With tweezers, she carefully removes the wafer-like filter and stuffs it into a test tube.
Back in her laboratory, she will extract DNA from the skin cells, feces, urine, and other bits of material left behind by aquatic inhabitants. The DNA is then run through assays to identify target species of fish, amphibians, snails, turtles, and other creatures. With that one sample, Goldberg can also detect rare and invasive species as well as disease-causing organisms like Bd and ranavirus.
As one of the world’s leading amphibian eDNA researchers, Goldberg analyzes more than a thousand samples each year from all over the world, including endangered frogs from the Panama forests visited by Pessier. She and her team have developed nearly 50 assays, each uniquely designed for a particular species.
Not only does eDNA improve and simplify the process of monitoring aquatic species, it’s also safer, more efficient, and minimally invasive. Now, instead of tromping through fragile wetlands—turning over rocks and kicking up mud, which can harm the animals living there—scientists can get answers with only a few water samples.
When Goldberg first learned of the concept as a graduate student in 2008, it transformed her world.
“As an ecologist, I spend a lot of time looking for rare species out in the field and not always finding them even though we know they are probably there,” she says. “When I heard about eDNA that detected amphibians in water, I was so excited. I knew it could have huge implications for managing and conserving rare species.”
By 2011, Goldberg had a contract with the Department of Defense to bring eDNA surveillance into the real world as a practical tool for wildlife conservation. Joined by fellow researchers Katherine Strickler and Alex Fremier in the School of the Environment, they set out to develop reliable techniques that would enable them to detect rare amphibians and fish on military bases across the United States.
“Our military bases are some of the last preserved parts of ecosystems that have otherwise been developed or plowed under,” Goldberg says. “They contain a lot of the nation’s endangered species. If you think about it, even a bombing range, for example, is much less disturbance to a salamander than is a shopping mall.”
She began the project by adapting protocols for working with poor-quality DNA that she’d learned as a doctoral student at the University of Idaho. In 2015, she joined WSU and designed her lab to use these new methods for processing eDNA samples.
Recently, Goldberg, Strickler, and wildlife biologist Jeff Manning in the School of the Environment were awarded another $1.4 million DoD contract to continue improving eDNA detection especially for species that are very rare and present in low numbers. They want to increase test sensitivity to handle some of nature’s most challenging conditions such as highly acidic water or very large ponds.
The biggest challenge for eDNA surveillance, however, may lie in the frontline battle to prevent a deadly salamander fungus from entering the United States and other vulnerable parts of the world.
In 2013, scientists were alarmed to discover massive salamander die-offs occurring throughout Europe from a new strain of chytrid fungus similar to Bd. Known as Batrachochytrium salamandrivorans, Bsal, or salamander chytrid, the disease is especially threatening to the United States, a global hotspot for salamander biodiversity.
Thanks to lessons learned during the frog Bd pandemic, the new infection was quickly identified and international barriers were established to prevent spread of the pathogen. By 2016, the U.S. Fish and Wildlife Service had banned imports of 201 salamander species.
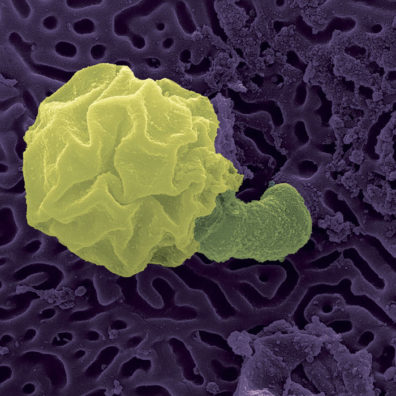
Jesse Brunner, associate professor in the School of Biological Sciences, is on the National Bsal Task Force and says the fungus has not yet been detected in North America.
“That’s really a good thing,” he says. “Allan Pessier, Caren Goldberg, and I are working on developing better approaches to screen animals and try to prevent it getting here. Millions of amphibians are imported into the U.S. every year, mostly through the pet trade.
“It’s very unregulated—we know Bd is found in some of these animals,” Brunner says. “We want to use eDNA testing to screen a whole shipment at a time rather than test each animal individually. The idea is that we can collect a handful of samples from the water and have a high probability of ensuring there isn’t infection in that group of animals.”
Worldwide surveys indicate these infectious fungi likely emerged from Asia where over millions of years, the local amphibian species developed a resistance to it.
“The exact origin may be uncertain but what is clear is that the movement of animals for the pet trade is moving pathogens like Bd and Bsal around the world,” says Brunner. “So, we can expect to see more emerging dangerous pathogens in the future rather than fewer.”
And while Asian frogs and salamanders seem to have a natural immunity, the fungus can wreak havoc when moved to a new location or into a novel species, he says. “That’s when you often see some of the worst outcomes.”
The Bd fungus is a devastating example. Brunner says frogs rely on their skin for breathing as well as electrolyte balance. When Bd invades skin cells, it disturbs the frog’s ability to regulate water and electrolytes, which leads to changes in the blood that essentially cause a heart attack.
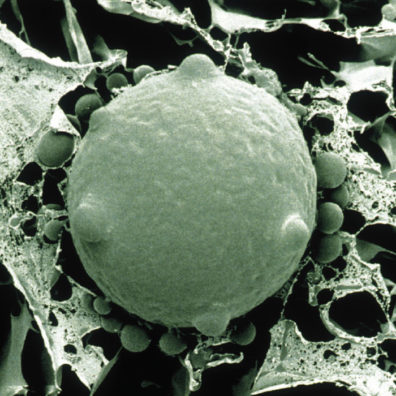
“It’s sort of like whole-body athlete’s foot that ends up killing them,” he says.
Though most salamanders breathe using both lungs and skin, it’s a similar story when they’re infected with Bsal—within days the fungus causes ulcers and sloughing tissue that lead to apathy, loss of appetite, and death. As one researcher put it, “It’s death by a thousand holes.”
Besides the fungus, Brunner is also concerned about one more “cold-blooded killer” called ranavirus that can cross-infect fish, reptiles, and amphibians.
“Ranavirus has a global distribution now,” he says. “It can be a really nasty infection—the virus gets into every bit of tissue they have, every cell, where it causes massive damage and organ failure. Thankfully, it doesn’t replicate at warm-blooded temperatures.”
The curious question is how some animals manage to control these viral and fungal infections so they don’t cause severe illness or death. Part of Brunner’s research is aimed at determining the factors that lead to this resistance and why catastrophic losses occur in some places and not others. He and his fellow scientists are following
several clues.
Erica Crespi, a physiologist and associate professor in the School of Biological Sciences, studies the way stress affects an amphibian’s early development. Frogs and salamanders are very sensitive to environmental changes which can trigger spikes in their stress hormone corticosterone.
“Just as in pregnant mammals where elevated stress hormones can cause premature birth, high corticosterone can shorten an amphibian’s development time and affect how the brain and lungs develop and cause other lifelong impacts,” she says.
Brunner says the idea that long-term chronic stress can suppress the immune system and make it harder for an animal to fight off infections has been studied by biologists for decades.
“In its simple form, the hypothesis says that anywhere we see human activities or other stressors, we should see big outbreaks of disease, but it’s not that simple. Stress doesn’t always translate into outbreaks.”
He and Crespi are trying to determine how individual animals respond to environmental stressors such as increased salinity or water temperature, and how that scales up to negative population outcomes like a pond full of floating frogs.
“The underlying stress mechanisms we’re studying apply to all sorts of animals like elk, fish, or any other species—and disease outbreaks in general,” says Brunner.
The investigation continues at WSU Vancouver, where Jonah Piovia-Scott, assistant professor in the School of Biological Sciences and a member of the National Bsal Task Force, is exploring the effects of climate change on chytrid fungal diseases.
“Neither Bd nor Bsal tend to do well when it’s hot,” he says. “So, some aspects of climate change may actually help amphibians with these pathogens. But other aspects may make them more susceptible. For example, if ponds dry up earlier in the season, it will decrease the amount of time amphibians have to develop. The stress will force them to develop faster, which may make them more susceptible to disease later in life.”
Piovia-Scott is often asked why we should care about amphibians and his answer is unequivocal.
“These amazing, beautiful, and wonderful organisms have intrinsic value, and are a part of our world we’re losing quite rapidly,” he says. “They are also integral components of the ecosystem—an important food source for some animals and they themselves eat insects, worms, and snails. Like salmon who are eaten by bears and fertilize the forest, amphibians are also an important link between aquatic and terrestrial systems.”
Indeed, isolated and far away, every frog and salamander die-off creates a domino effect that ultimately impacts the planet. Streams that were once crystal clear turn green without tadpoles to eat the algae. Human infections like malaria and dysentery spread more rapidly without amphibians to eat mosquitos and flies.
“It’s a very good example of how small the world has gotten,” says Pessier, who also specializes in biosecurity and reintroduction programs for endangered species.
“Diseases like Bd and Bsal are moved around by people. Domestic cows don’t move from Asia to the U.S. without a huge number of diagnostic tests. But for frogs, you just need the right permits and you can move them all over the world without concerns about disease.
“Once Bd has moved through an area, the amphibian biodiversity drops to virtually nothing and there is no way to mitigate the fungus in the wild,” he says. “So, the last resort strategy is to develop survival assurance populations (SAP). We capture threatened species to preserve their genetic diversity and then try to breed and maintain a colony in captivity until they can be reintroduced to the wild, once we have a way to deal with the fungus.”
Pessier works with SAP in Madagascar, Ecuador, Panama, and many other areas around the world to diagnose disease issues such as vitamin A deficiency in captive Panamanian golden frogs.
Closer to home, he is joining Crespi and Goldberg to protect Washington’s last surviving remnant of northern leopard frogs in the Columbia Basin. Working with the Washington Department of Fish and Wildlife, they hope to reintroduce and expand populations within the state.
Their intentions clearly reach beyond academia to a deeper love of the Pacific Northwest and our amphibian wildlife. Drafted in the spirit of Teddy Roosevelt, their proposal reads, “Do what you can, with what you have, where you are.”
It’s a philosophy all five faculty members stand behind. Their shared interests and mutual support have multiplied efforts to protect frogs and salamanders throughout the world.
“Conservation is an interdisciplinary science,” Crespi says. “Having Jesse, Caren, Allan, and Jonah here allows me to do projects I could never do in isolation.”
Read more
The legacy lives on — Doctoral student Erim Gómez works with associate professor Rodney Sayler in the WSU Endangered Species Lab to conduct the first comprehensive survey of frogs and salamanders on the Palouse Prairie since the 1930s.
Guide to Amphibians and Reptiles of Washington (Burke Museum)





